Optimizing AAV Plasmid Preparation and ITR Sequencing
The adeno-associated virus (AAV) is a powerful vehicle for gene therapy; however, working with AAV vectors can be challenging. Each construct contains two inverted terminal repeat (ITR) sequences, usually 145 bp in length, that flank the gene of interest (see figure below). The ITRs form highly stable T-shaped hairpins, which are critical for the replication and encapsidation of viral DNA. These structural features can also wreak havoc on traditional cloning workflows. Spontaneous deletions in the ITR region plague DNA preparations of the transfer plasmid, and the formation of secondary structure stymies confirmatory sequencing. Hence, investigators are hit with a double whammy: not only are AAV vectors prone to ITR truncations, but also such mutations are tricky to detect. Here, we’ll discuss the challenges of propagating and validating AAV plasmids and propose an optimized approach to the workflow.
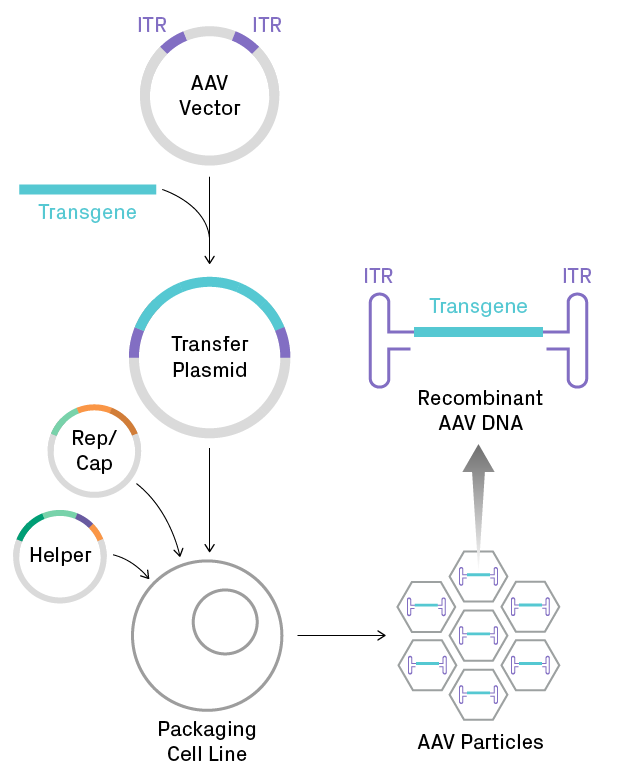
Production of recombinant AAV particles. A gene of interest is cloned into an AAV vector to create a transfer plasmid, which is then introduced into a eukaryotic cell line. Generating functional viral particles requires co-transfection with several viral genes, supplied by packaging plasmids (i.e., Rep/Cap and helper plasmids). Recombinant single-stranded DNA, containing the transgene flanked by inverted terminal repeat (ITR) sequences, is packaged into capsids. The ITRs form very stable hairpins, which are problematic for amplification and sequencing of AAV plasmids during the early steps of the workflow.
ITR Instability
The ITR regions in AAV plasmids are notoriously unstable. Their palindromic nature and high GC content make them prone to full or partial deletions during propagation in bacteria1. As a result, a significant portion of clones after transformation may contain mutated ITRs, and plasmid DNA preparations from a liquid culture may contain a mixed population (see figure below). The underlying mechanism of this phenomenon in bacteria is not well understood; secondary structure and strand slippage in the replication fork may play a role2. Since the loss of ITR sequences confers a growth advantage to the host bacteria, a mutation can rapidly amplify in a growing culture1. Even with the best standard practices, AAV plasmid preparations can contain a significant fraction (5-15%) of plasmid DNA with mutated ITR sequences3. Disruption of ITRs can lead to reduced efficiency in viral packaging and increased variability in downstream experiments4.
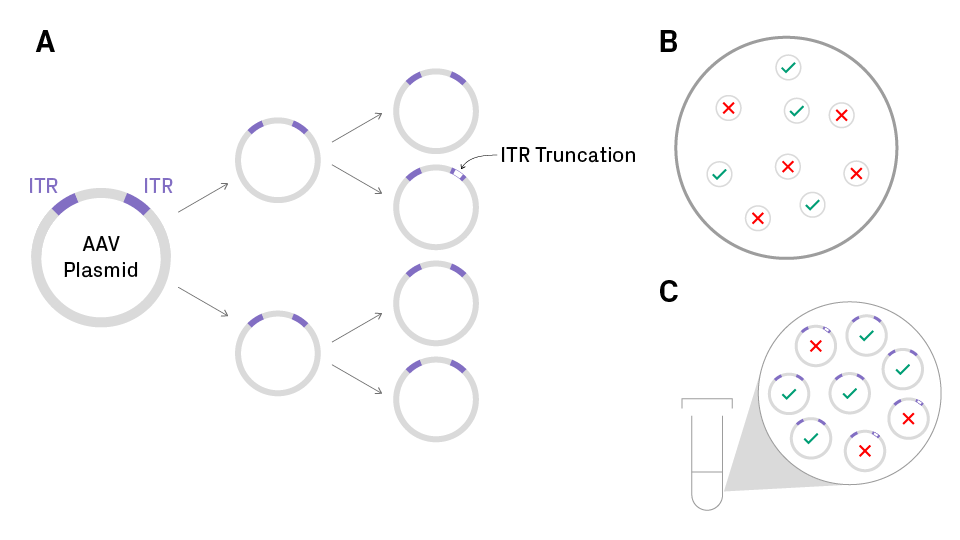
Plasmids with ITR sequences exhibit high instability. (A) Mutations and truncations frequently arise in the ITRs during propagation in bacterial cells. (B) When selecting colonies from an agar plate, many clones may contain the mutation. (C) When growing bacteria in liquid culture, the resulting plasmid preparation may contain a mixture of DNA molecules.
Challenges in Detecting ITR Mutations
Given the frequency of truncations that arise in AAV plasmids, it’s critical to monitor the integrity of ITRs throughout cloning, especially prior to viral packaging. Sanger sequencing is the gold standard for plasmid sequence confirmation; however, standard protocols fail to read through ITR regions. The strong secondary structure of ITRs inhibits DNA polymerization, leading to a significant drop in signal intensity in chromatograms (see figure below). Common strategies for sequencing difficult templates, such as adding DMSO and/or betaine, are ineffective for ITRs5.
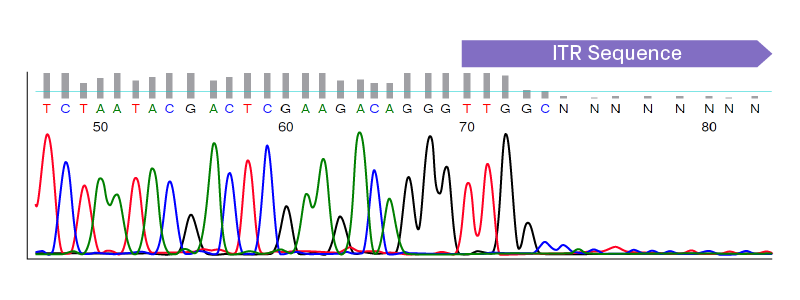
Sequencing chromatogram showing early termination of signal at the start of the ITR sequence. Standard protocols for Sanger sequencing fail to read through ITR regions.
Restriction digestions are traditionally performed to screen for ITR deletions. The enzymes SmaI and SrfI are often used, as they cut within the C-C’ arm of the ITR3. Given the limited resolution of gel electrophoresis, diagnostic digests fail to detect small deletions and point mutations outside the restriction site (see example below). In short, restriction enzyme analysis alone is insufficient to monitor ITR integrity in plasmids.
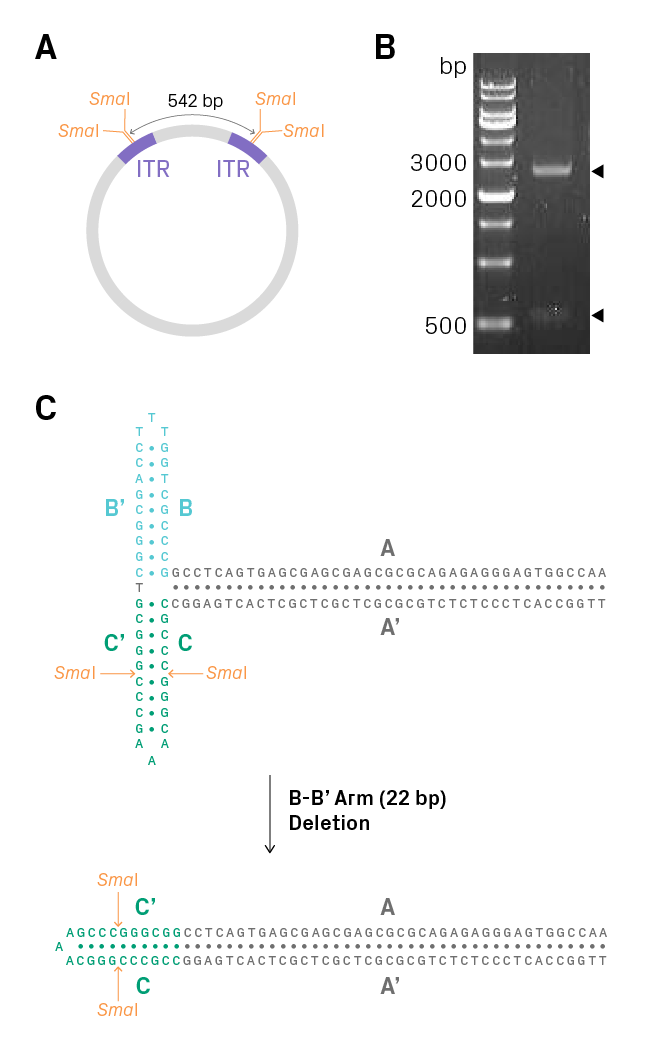
Restriction enzyme analysis fails to detect small deletions in ITRs. (A) Restriction map of an AAV plasmid in which SmaI cuts twice within the ITR region. (B) Gel of SmaI digestion showing bands of expected size. (C) The analyzed plasmid preparation actually has a deletion of the B-B’ arm in one of the ITR regions, discovered through GENEWIZ’s AAV-ITR sequencing. Agarose gel electrophoresis does not have sufficient resolution to identify a loss of 22 bp.
How to Optimize AAV Plasmid Preparation
The following proactive measures can minimize the risk of mutations in ITRs and provide effective quality control of AAV plasmids preps.
1. Use a bacterial strain that maintains ITR integrity
Recombination-deficient strains (e.g. JC8111, SURE2, Stabl3, XL10-Gold) are typically used when cloning AAV constructs, as they reduce the likelihood of ITR deletions1. However, depending on the particular construct, it can be very difficult to isolate a clone with fully intact ITRs using commercial strains, even those lacking RecA. To address this shortcoming, GENEWIZ developed proprietary bacterial strains specifically optimized to maintain ITR integrity during plasmid propagation. The GENEWIZ strains outperformed three commercial strains in the testing of eight AAV plasmids with different backbones. This difference was more pronounced for the three most challenging plasmids, with commercial strains failing to generate a clone with fully intact ITRs for at least two of the plasmids tested.
With suboptimal bacterial strains, plasmid preparation becomes more tedious: you need to screen more colonies, assuming a correct clone can be obtained at all. Also, during scale-up, there’s a greater chance that a mutation will arise, resulting in a mixed population of plasmids that can affect downstream experiments or require reisolation of a clone.
2. Use a robust, sensitive assay to detect ITR mutations
As discussed above, standard sequencing protocols are ineffective in reading through ITRs, and restriction digestions provide limited information about ITR sequences. The lack of adequate methods to measure ITR integrity often leaves researchers uncertain about the quality of their AAV plasmid preparations. In response, GENEWIZ developed a radically new Sanger sequencing protocol that can read through the challenging ITR regions, providing confirmation of ITR integrity at single-nucleotide resolution (see figure below).
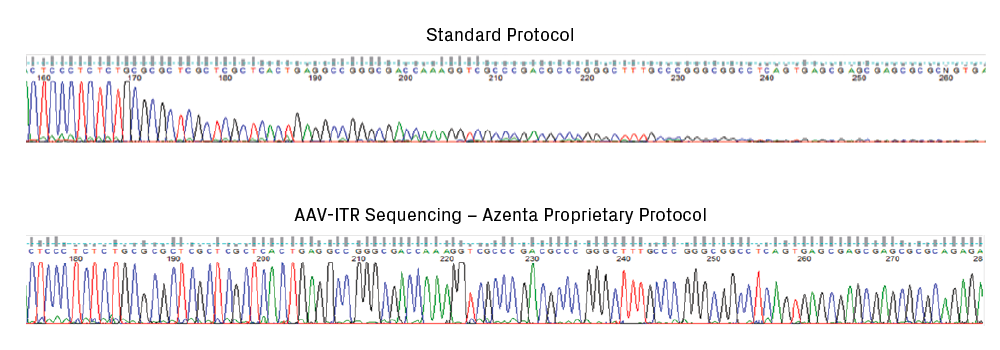
An ITR region analyzed using a standard Sanger sequencing protocol compared to GENEWIZ’s AAV-ITR sequencing method. The latter maintains a strong signal through the ITR sequence, providing a useful read.
Some truncated ITRs are surprisingly more challenging to sequence than intact ones, as they lead to hairpin structures with even greater stability. Alternative sequencing protocols may have some success reading through intact ITR regions but will likely fail to sequence these more difficult truncations. Consequently, the presence of such a mutation in a mixed population can escape your notice. The GENEWIZ AAV-ITR sequencing method is sufficiently robust to read through both wild-type and mutant ITRs, enabling detection of very challenging ITR truncations in clonal and heterogeneous populations of plasmid DNA.
3. Perform QC checks for ITR integrity early and often
Catching mutations early and taking immediate corrective measures can save substantial time and effort down the line and lead to more reproducible results in later experiments. A robust AAV plasmid preparation workflow includes multiple QC checks (see figure below). Since ITR truncations can arise at any step involving amplification in bacteria, vigilance is key to ensuring ITR integrity throughout the cloning process. It’s important to analyze plasmids with AAV-ITR sequencing after each DNA preparation to confirm that ITRs are intact and clonal. For preps showing a mixed population, a clone with correct ITR sequences should be reisolated, preferably in a bacterial strain that is optimized for AAV constructs.
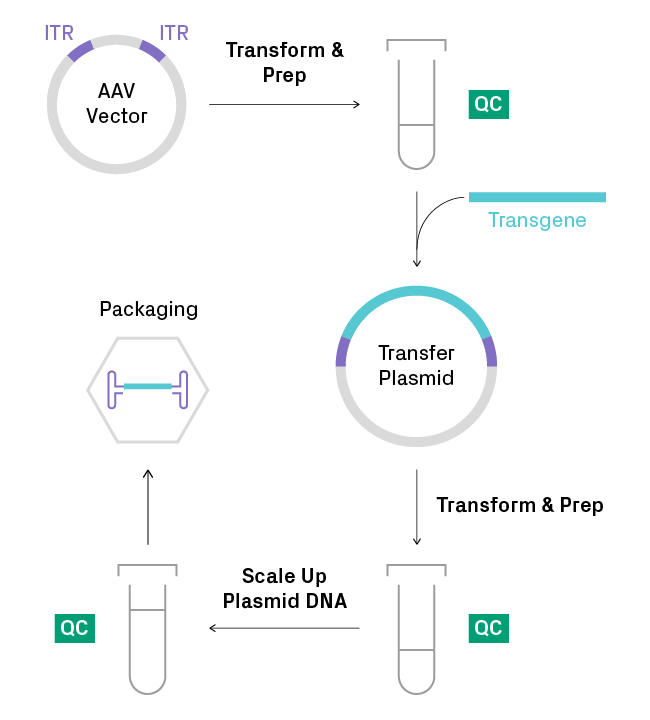
Recommended quality control (QC) checkpoints for ITR integrity. The ITR regions should be analyzed by AAV-ITR sequencing after each plasmid DNA preparation to ensure the ITRs are fully intact and clonal.
Key Takeaways
- The inverted terminal repeat (ITR) sequences of AAV vectors are highly unstable, often accumulating deletions during plasmid propagation in bacteria
- Standard methods for plasmid sequence confirmation are inadequate for detecting ITR truncations that can negatively impact downstream experiments
- Using the latest technologies for AAV plasmid preparation and sequencing can significantly expedite research and improve the quality of recombinant AAV DNA
Conclusion
The structural features of ITR sequences pose significant quality control issues for investigators aiming to build and clone AAV constructs. However, recent technological advancements have addressed the most difficult challenges, enabling amplification and confirmation of fully intact ITR regions.
To learn more, read our tech note “An Efficient and High-Fidelity Approach to AAV Plasmid Preparation”.
References
1. Wilmott, P., Lisowski, L., Alexander, I. E. & Logan, G. J. A User’s Guide to the Inverted Terminal Repeats of Adeno-Associated Virus. Human Gene Therapy Methods 30, 206–213 (2019).
2. Oliveira, P. H., Prather, K. J., Prazeres, D. M. F. & Monteiro, G. A. Structural instability of plasmid biopharmaceuticals: challenges and implications. Trends in Biotechnology 27, 503–511 (2009).
3. Gray, J. T. & Zolotukhin, S. Design and Construction of Functional AAV Vectors. in Adeno-Associated Virus 25–46 (Humana Press, 2011).
4. Savy, A. et al. Impact of Inverted Terminal Repeat Integrity on rAAV8 Production Using the Baculovirus/Sf9 Cells System. Human Gene Therapy Methods 28, 277–289 (2017).
5. Kieleczawa J. Fundamentals of sequencing of difficult templates–an overview. J Biomol Tech 17(3), 207–217 (2006).sequencing is the best solution.
Daily Insight
Recent Posts
Recent Post Tags
- next generation sequencing
- gene synthesis
- rna sequencing
- transcriptomics
- sanger sequencing
- Event
- GENEWIZ Update
- aav-itr
- cell and gene therapy
- itr sequencing
- mRNA
- proteomics
- aav plasmid prep
- antibody discovery
- genomics
- grant winner
- olink
- quantitative pcr (qPCR)
- rna therapeutics
- single-cell sequencing
- PacBio
- aav
- circular rna (circRNA)
- digital pcr (dPCR)
- immunity
- lentivirus
- long-read sequencing
- oxford nanopore sequencing (ont)
- pcr + sanger sequencing
- whole genome sequencing
- whole plasmid sequencing